RESEARCH
Our research interests are at the interface of chemistry and biology and focus on the use of synthetic organic chemistry to develop molecular tools to enable the study of biological systems. Our research programmes can be grouped into three main areas, (1) investigation of epigenetic changes in health and disease; (2) novel probes to measure and target cellular REDOX; and (3) targeting infectious diseases.
References
- M. Brand, et al. 2020, ChemRxiv. Preprint. https://doi.org/10.26434/chemrxiv.12081999.v1.
- M. Schiedel, M. Moroglu, D. M. H. Ascough, A. E. R. Chamberlain, J. J. A. G. Kamps, A. R. Sekirnik, S. J. Conway Angew. Chem. Int. Ed. 2019, 58, 17930.
- A. R. Sekirnik (née Measures), D. S. Hewings, N. H. Theodoulou, L. Jursins, K. Lewendon, L. E. Jennings, T. P. C. Rooney, T. D. Heightman, S. J. Conway. Angew. Chem. Int. Ed., 2016, 55, 8353.
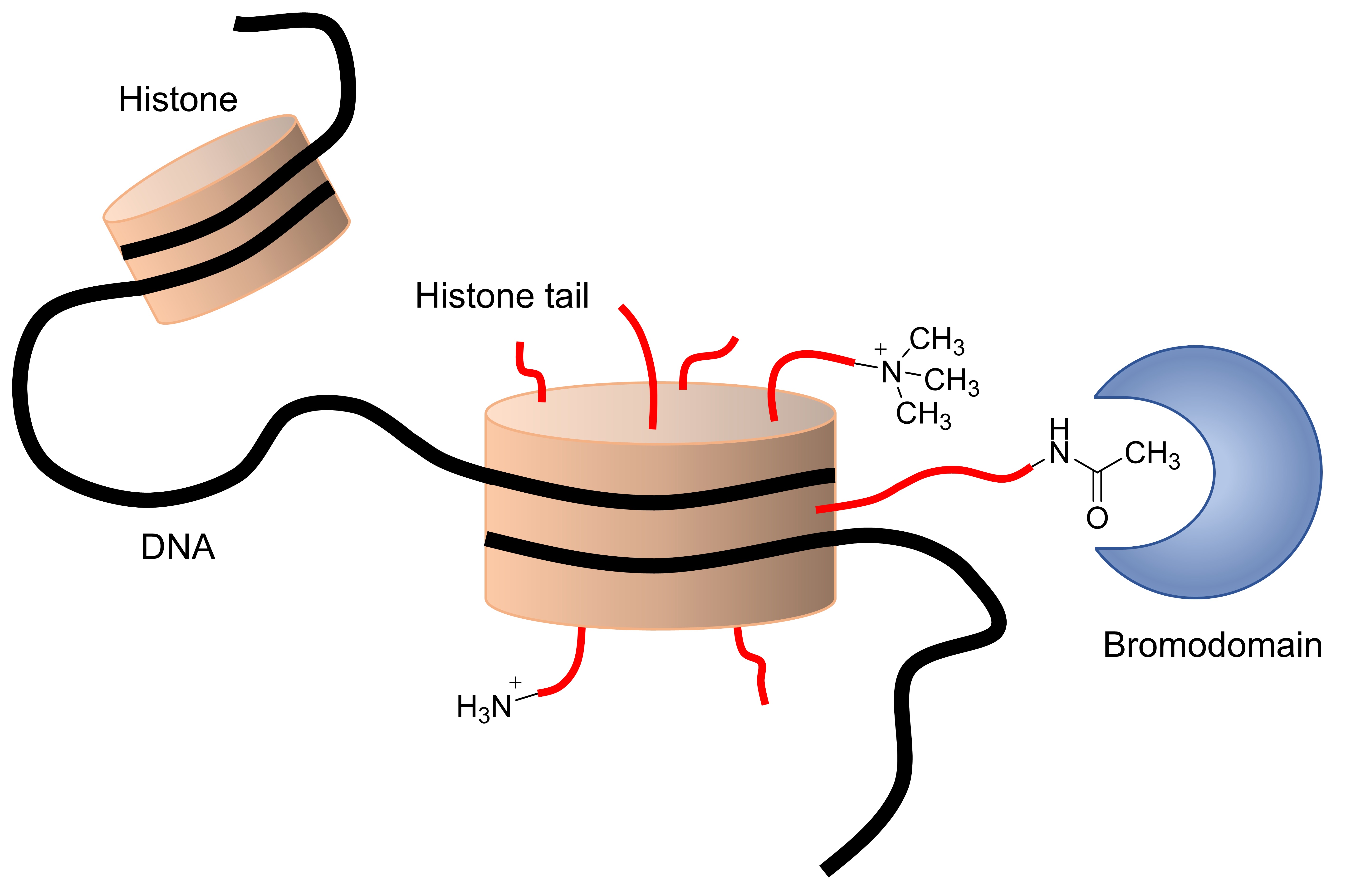
Investigation of epigenetic changes in health and disease
Epigenetics is the study of heritable changes in gene expression that do not entail a change in DNA sequence, or, how phenotype is linked to genotype. Epigenetic modifications occur regularly and are essential for fundamental biological processes such as cell differentiation to form different tissues like skin, brain, or heart. However, changes in the epigenome can also lead to undesired effects and the cause of diseases including cancer.
Histone modifications are key elements of epigenetics. For instance, lysine residues can be methylated or acetylated by so-called writer proteins. Epigenetic erasers reverse these processes, and bromodomains are epigenetic readers of acetylated lysines.
We are interested in the development of chemical probes for epigenetic proteins including those with bromodomains. Using these chemical tools, we can explore their biological function and phenotypical consequences of chemical intervention, helping to develop novel therapeutic strategies for diseases including cancer.
References
- O’Connor, L. J. et al. ACS Cent. Sci. 2017, 3, 20–30.
- Collins, S. L., Saha, J., Bouchez, L. C., Hammond, E. M. & Conway, S. J. ACS Chem. Biol. 2018, 13, 3354–3360.
- L. J. O’Connor, C. Cazares-Körner, J. Saha, C. N. G. Evans, M. R. L. Stratford, E. M. Hammond, S. J. Conway. Nature Protocols, 2016, 11, 781.
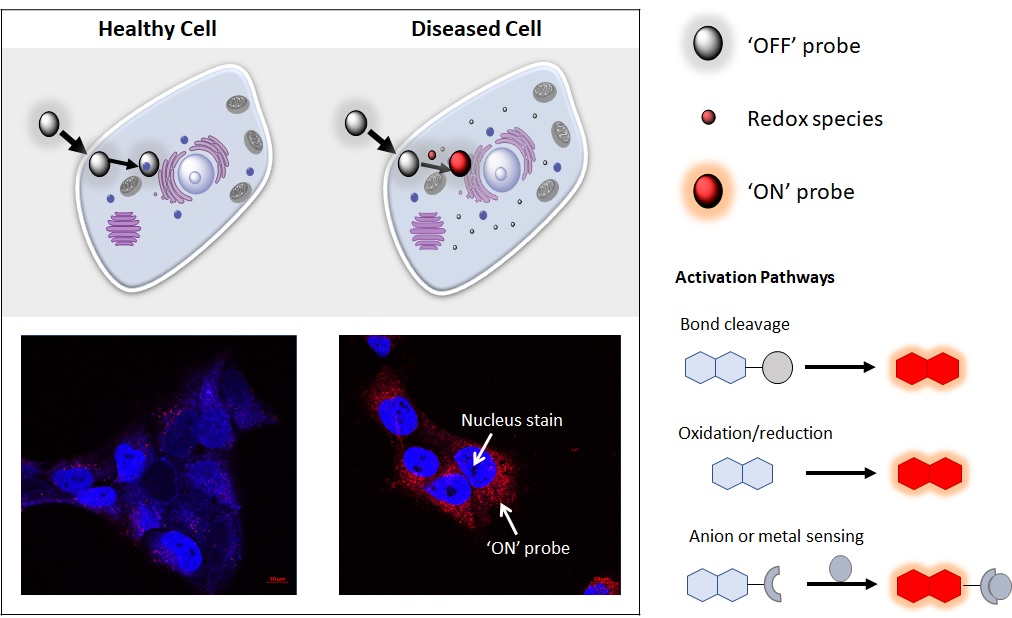
Novel probes to measure and target cellular Redox
In collaboration with Professor Ester Hammond, Oxford Institute for Radiation Oncology, Department of Oncology, University of Oxford
The cellular environment within a living system is thought to span a spectrum between reducing and oxidising conditions. Reducing conditions are characterised by lack of oxygen and chemicals which contain hydrogen. Oxidising conditions are described by the presence of oxygen and reactive oxygen containing species. The reducing and oxidising species in a cellular environment are responsible for the overall Redox state. Redox species are involved in multiple cellular processes including cell signalling and metabolism. Redox equilibrium is important for normal cell function. Perturbation of the Redox environment can give rise to cellular changes that result in diseases such as cancer. Our research involves the development of chemical tools which utilise optical and radio-imaging techniques; to provide real time information on changes in the Redox environment. Innovative tools will provide a platform to yield novel information on fundamental biological processes and new ways to predict or diagnose disease.
References
- M. Brand et al. ACS Chem. Biol. 2015, 10, 22.
- L. E. Jennings, A. R. Measures, B. G. Wilson, S. J. Conway. Future Med. Chem., 2014, 6, 179.
- J. Healy et al. Biochemistry, 2014, 53, 1982.
Targeting infectious diseases
Parasites are the causative agents of Chagas disease, leishmaniasis and schistosomiasis, three major neglected tropical diseases (NTDs) that affect millions of people in some of the poorest communities in the world. The parasites (Trypanosoma, Leishmania and Schistosoma) exist in different forms in their life cycle to adapt to their changing habitats, which include the vector and the host bloodstream. We are investigating whether epigenetic mechanisms enabling changes in gene expression work to regulate the life cycle of parasites, and ensure their survival. Bromodomains, “readers” of lysine acetylation (KAc), have emerged as key gene expression regulators and a promising new class of drug target in humans. Given bromodomain-containing proteins (BCPs) play a fundamental role in regulating transcription in humans, we hypothesise that they will play equally important roles in parasites. We are investigating whether these proteins can be novel therapeutic targets to impede the life cycle of parasites and open new avenues for the treatment of infectious diseases.
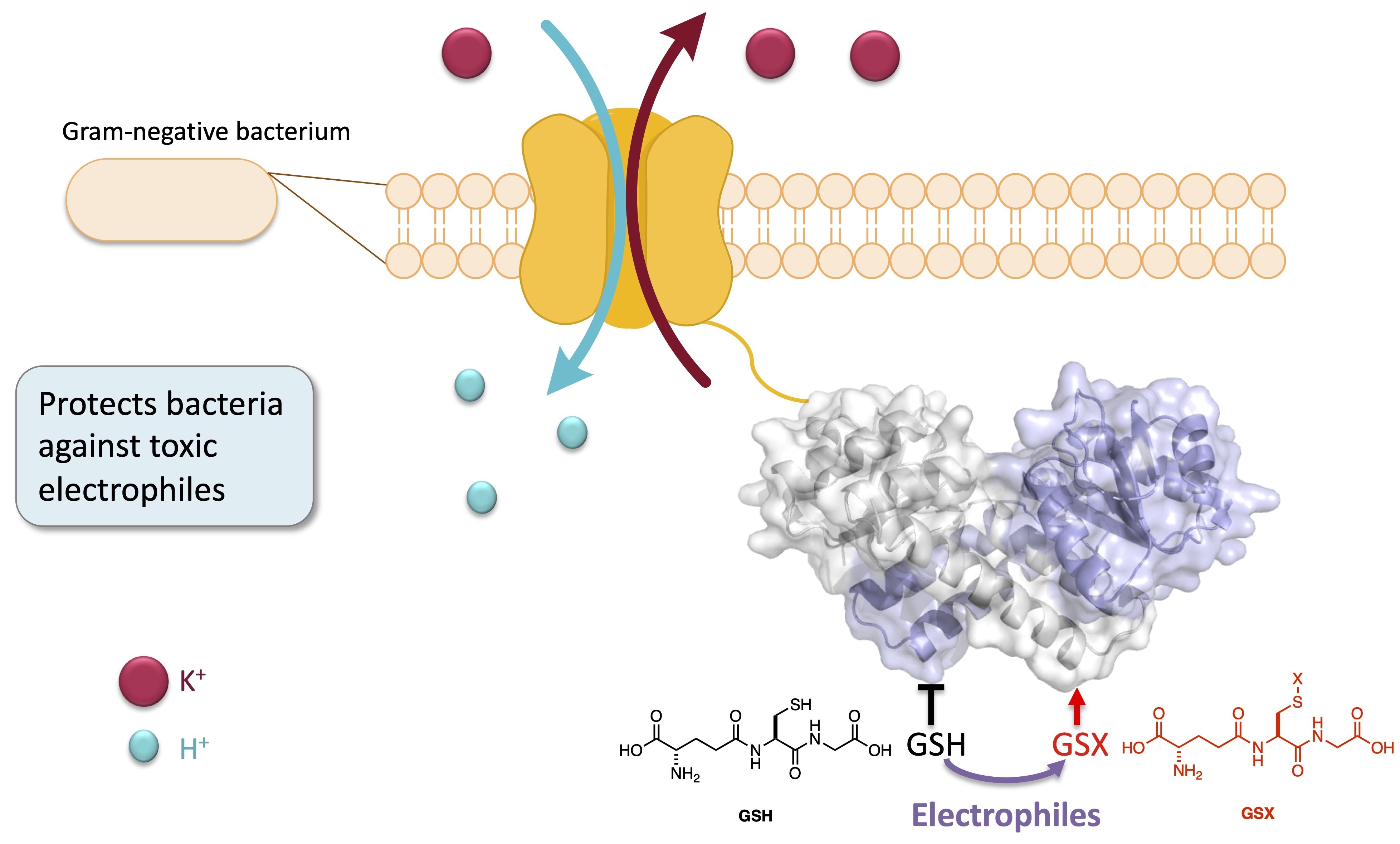
We are also interested in developing compounds to probe new antibiotic targets. The world is about to reach an antibiotic global crisis, and therefore it is vital to develop new antibiotics. Of particular interest are the ESKAPE pathogens, which are the leading cause of hospital acquired infections. We are aiming to probe whether Kef, a bacterial proton/potassium antiporter, is of interest as an antibacterial target (see figure). Previous work done by the group has shown glutathione-X-conjugates are able to bind and activate Kef causing inhibition of cell growth. We are currently developing peptide-mimetic ligands capable of binding and activating the Kef system.